In the rapidly evolving field of genomics, employing NGS Library Preparation Kits effectively is crucial for successful sequencing. These kits streamline the process of preparing DNA or RNA samples, allowing researchers to focus on analysis rather than procedures. Proper use of these kits not only enhances data quality but also increases throughput in laboratory settings.
Understanding the intricacies of NGS Library Preparation Kits can be challenging. Researchers often encounter issues such as sample degradation or adapter compatibility. This highlights the importance of following protocols closely while also allowing room for experimentation. A balance between adherence and innovation is key.
Being familiar with specific tips can dramatically improve outcomes. For example, optimizing enzyme concentrations and understanding fragmentation methods are vital. It’s essential to reflect on your approach and adapt as necessary, ensuring reliable and reproducible results. These considerations foster a deeper understanding of the technology and its applications in various research fields.

Next-Generation Sequencing (NGS) library preparation is a vital step in genomic research. Understanding the basic concepts is crucial for accurate data generation. Effective library preparation leads to high-quality sequencing results. This process involves fragmenting DNA, ligating adapters, and amplifying the resulting library. Each of these steps must be meticulously performed to avoid errors.
One tip for effective NGS library preparation is to ensure optimal DNA fragmentation. Too large or too small fragments can affect your sequencing results. Aim for a balanced size distribution. Another important factor is to maintain a clean environment. Contamination can introduce variability and compromise your results.
It’s essential to validate the quality of your library before sequencing. Using genome analysis techniques can provide insights into the library's concentration and fragment size. Reflect on your techniques and adjust where necessary. Sometimes, even slight deviations can lead to significant differences in outcomes. Keeping detailed records during this process can help in troubleshooting issues and ensuring reproducibility.
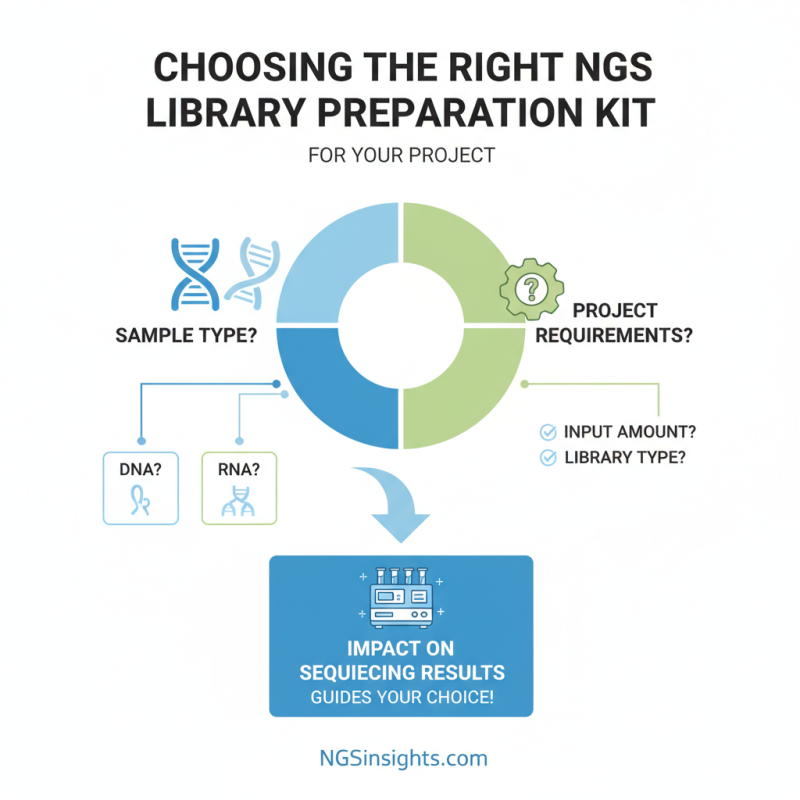
Choosing the right NGS library preparation kit for your project is crucial. A good kit can significantly impact your sequencing results. Start by considering the specific requirements of your project. What type of samples are you using? Are they RNA or DNA? The answers guide your choice.
Pay attention to the throughput and recovery rates of the kits. If you are working with limited samples, select a kit that maximizes yield. Also, review the protocol complexity. Some kits require advanced skills, while others are straightforward. A complicated process may lead to errors, affecting data quality.
Lastly, consider the cost versus benefits. While cheaper kits might save money, they may not meet your project’s needs. Weigh the trade-offs carefully. Review literature or seek feedback from peers who have used specific kits. This helps ensure you make an informed decision. Whatever kit you choose, always have a backup plan ready.
Library preparation for next-generation sequencing (NGS) is a critical step in genomic research. Following a step-by-step protocol enhances efficiency. A well-executed library preparation can improve sequencing yield and data quality. According to industry reports, poor library preparation can lead to significant sample loss—up to 30% in some cases.
One essential aspect is to optimize sample quality. Impurities can inhibit enzyme activity, affecting the final library. It is crucial to assess DNA integrity using methods such as gel electrophoresis or bioanalyzer systems. Additionally, accurate quantification of your samples will ensure the right amount of material is used. Moreover, adhering to temperature guidelines during reaction setups can mitigate enzymatic degradation. Observing these parameters prevents inconsistencies and data flaws.
Another critical point is the choice of adapter sequences. Not all adapters fit every application. Some experiments may require specific modifications or indexing strategies for multiplexing. This customization should be reflected in your protocols. Reflecting on failed experiments shows that a one-size-fits-all approach may not be effective. Continuous learning and protocol adjustments are necessary for achieving optimal results in NGS library preparation.
When working with NGS library preparation kits, common pitfalls can impact the quality of your results. One common issue is inconsistent pipetting. Ensure that you use calibrated pipettes for accurate measurements. Small volume discrepancies can lead to significant variations in library size and yield.
Another frequent mistake is neglecting to adjust for sample quality. Low-quality RNA or DNA can lead to poor library performance. Always perform a quality check before starting the library prep process. This small step helps prevent wasting reagents and time.
Consider the storage conditions for your kits. Improper storage can degrade enzymes and reagents. Store them according to manufacturer instructions. Environmental factors can greatly influence shelf life. A little attentiveness here can ensure optimal performance during library prep.
The final tip: be mindful of contamination. Use dedicated equipment and work in a clean environment. Cross-contamination can easily compromise your results. Implementing these steps can dramatically improve your library preparation efficiency.
| Tip | Description | Common Pitfall | Solution |
|---|---|---|---|
| 1. Thoroughly Read Protocols | Understanding the full protocol ensures correct application of the kit. | Skipping steps due to misunderstanding. | Spend time ensuring protocol is fully understood before starting. |
| 2. Maintain Clean Environment | Use a dedicated clean workspace to prevent contamination. | Contaminated samples leading to unreliable results. | Regularly clean workspace and use sterile tools. |
| 3. Use Proper Quantification Techniques | Reliable quantification of DNA/RNA is crucial for effective library preparation. | Inaccurate quantification leading to over or under loading. | Utilize recommended methods like Qubit or Bioanalyzer. |
| 4. Pay Attention to Temperature | Temperature sensitivity affects enzyme activity. | Enzyme degradation leading to inefficient processes. | Keep reagents at appropriate temperatures as stated in protocols. |
| 5. Verify Reagent Expiry Dates | Expired reagents can negatively affect library quality. | Using expired reagents resulting in failure of the library. | Regularly check and rotate stock based on expiry dates. |
| 6. Follow Proper Pipetting Techniques | Precision in pipetting prevents sample loss and variation. | Incorrect volumes leading to suboptimal reactions. | Practice consistent pipetting techniques; consider using multichannel pipettes. |
| 7. Use Fresh Solutions | Certain solutions degrade quickly; fresh solutions enhance reaction efficiency. | Using outdated solutions resulting in reduced yield or quality. | Prepare fresh solutions as indicated in the protocol before use. |
| 8. Optimize Fragment Size | Proper selection of fragment size improves sequencing outcomes. | Improper sizing can lead to suboptimal sequences. | Use size selection methods as recommended for the kit. |
| 9. Document Everything | Keeping detailed records helps in troubleshooting issues. | Loss of valuable data due to lack of documentation. | Create a standardized log for all steps taken during preparation. |
| 10. Calibrate Equipment Regularly | Regular calibration ensures reliability of equipment used in preparation. | Inaccurate results due to miscalibration. | Establish a regular calibration schedule for all equipment. |
Quality control (QC) in NGS library preparation is crucial. Effective QC measures ensure reliable sequencing results. A good starting point is to monitor the DNA input quantity accurately. Too much input can lead to adapter dimers. Too little can result in poor library diversity.
Employing quantitative PCR (qPCR) can provide insights into library quality. This technique helps detect the presence of the desired amplicon sizes. However, relying solely on qPCR might not always reveal all issues. It's essential to perform size selection using an instrument like a Bioanalyzer. This method effectively assesses fragment sizes, but it requires careful calibration.
Another consideration is the importance of preparing negative controls. Including these controls helps identify contamination or other issues early on. Results can be misleading without them. Review your workflow and identify potential sources of contamination. Optimize your preparation protocols based on observed challenges. Quality control is an iterative process, and mistakes can happen. Staying mindful of these factors allows for continual improvement in NGS library preparation.
This chart shows the effectiveness of various quality control measures used in Next-Generation Sequencing (NGS) library preparation. The data reflects the percentage of successful libraries generated under different quality control methodologies.